New Publication by Henrik Huizinga and Rosalie van Dijken in the International Journal if Pharmaceutics
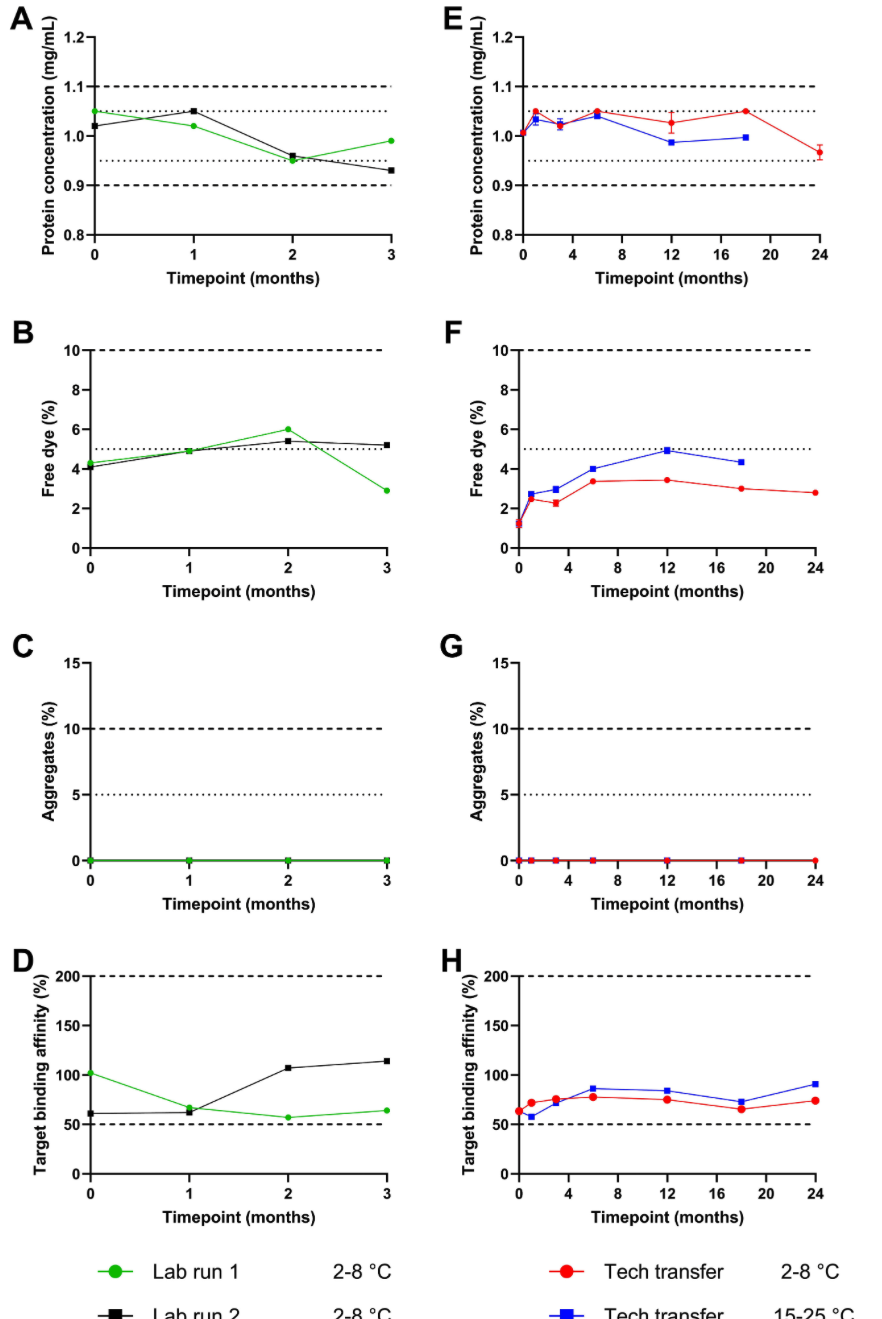
Our latest publication by Henrik Huizinga and Rosalie van Dijken et. al. entitled “Roadmap for the accelerated development and clinical translation of fluorescent tracers: Adalimumab-680LT as a proof of concept” can be found here.
Abstract
Keywords: accelerated drug development, fluorescent labelled antibodies, adalimumab-680LT
