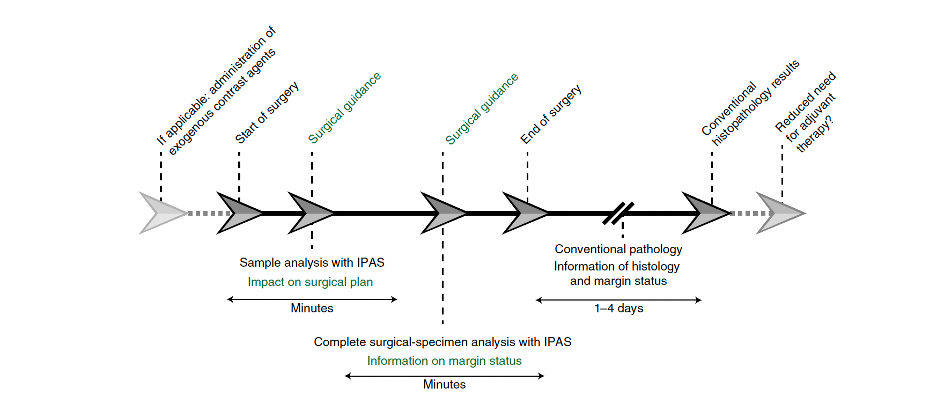
Intra-operative pathology assisted surgery (IPAS)
Removing all cancer tissue is one of the most important outcome parameters in oncologic surgery. However, no clinical tools are available during surgery that can assist the surgeon in real-time. To date, the surgeon receives the pathology results from the pathologist a few days after the surgery has been completed, thereby missing the opportunity to directly act on the obtained surgical excision results. Implementing novel optical imaging techniques for the enhanced analysis of the excised specimen during surgery can improve intra-operative clinical decision-making.
Focus
In surgical oncology, the ideal surgical outcome is the resection of the entire tumor bulk and the preservation of the adjacent (healthy) tissue. However, the occurrence of tumor-positive surgical margins remains high. The current standard of care for intraoperative surgical guidance is the fresh frozen sectioning (FFS) of tissue biopsies. Although helpful in some cases, this technique usually requires up to 1h and is only available for the analysis of small tissue biopsies. Unfortunately, the reliability of FFS for margin assessment is controversial, as it is susceptible to sampling error. Also, the technical quality of FFS is low compared to definitive histopathology.
Moreover, margin assessments of the entire excision specimen can take up several days, which precludes the possibility of altering the initial treatment plan or of intervening immediately during the initial surgery. When the final histopathology results are available, a second treatment (if possible at all because of typically altered anatomy and distortion of the initial anatomical orientation) often leads to overtreatment. This can involve mutilating surgical procedures or chemoradiotherapy to correct for incomplete initial surgery.
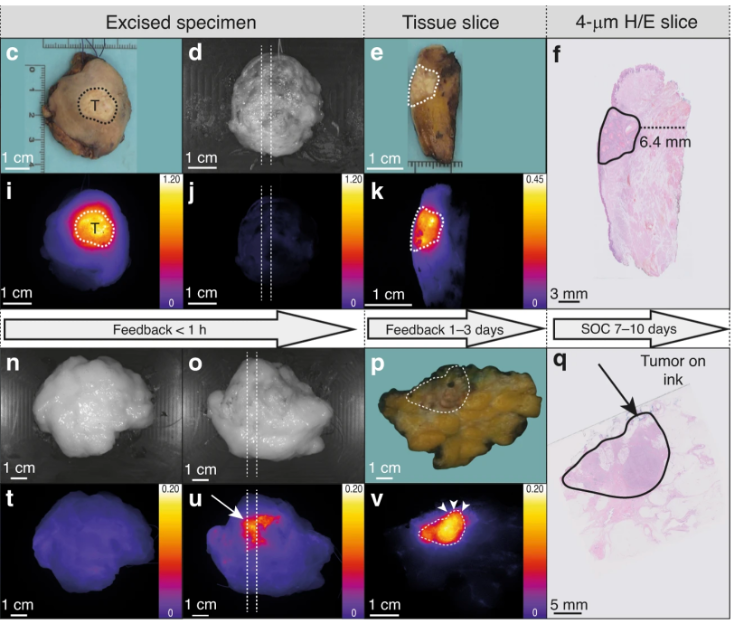
Real-time collaboration
The need for real-time information has led to increasing interest in intraoperative in vivo imaging techniques that can guide surgeons during a surgical procedure by providing information that the human eye cannot detect. However, the use of in vivo imaging techniques is constrained by regulatory concerns inherent to a sterile working environment. Also, variations in image-acquisition settings related to the surgical working area, and the absence of standardization and calibration protocols for existing imaging systems, have resulted in a lack of benchmarking and of quality controls.
These limitations can be redressed through the pathological assessment of excised tissue immediately after resection while the patient is still anaesthetized, which still allows for additional intraoperative intervention if needed.
Within the OMIG we focus on how the intraoperative implementation of novel optical imaging techniques (i.e. intraoperative pathology-assisted surgery [IPAS])—that is, the real-time collaboration between surgeons and pathologists—could affect surgical treatment strategies.