Diagnostic Accuracy Study Of Indocyanine Green For Perfusion Assessment Of The Parathyroid Glands In Thyroid Surgery (Pre-CLOVER)
One of the most common complications of thyroid surgery is postoperative hypoparathyroidism, transient or persistent, affecting up to 38% of patients. [1,2] During the procedure, it regularly happens that the parathyroid glands are damaged and are therefore no longer functional postoperatively. In 2016, the first study using ICG angiography to assess parathyroid vascularization was published. In this current study, we aim to optimize the standardization and quantification of this technique.
Background
The high complication rate is attributed to the fact that the parathyroid glands are difficult to spot in the wound area, due to their very small size (sometimes only the size of a pinhead) and the fact that it sometimes resembles adipose or lymph node tissue. Nevertheless, it is useful to know peroperatively whether the parathyroid glands are intact. If it appears that the parathyroid glands are insufficiently vital, it is possible to auto-transplant one of these parathyroid glands into a highly perfused tissue, such as the sternocleidomastoid muscle. The expectation is that the function of this parathyroid gland will eventually become sufficient again to prevent the patient from having to continue taking medication. [3] However, this is not necessary if the parathyroid glands appear to be sufficiently preserved during surgery. A review of our study group from 2022 found that the aforementioned studies are heterogeneous regarding the conclusion whether the use of ICG angiography can prevent postoperative hypoparathyroidism. This appears to be due to a total lack of standardization of the different protocols used, which involved many different cameras and doses of ICG. Also within most studies there seemed to be no attention for standardization of the protocol and also quantification of the fluorescence signal found. [4] (Figure 1)
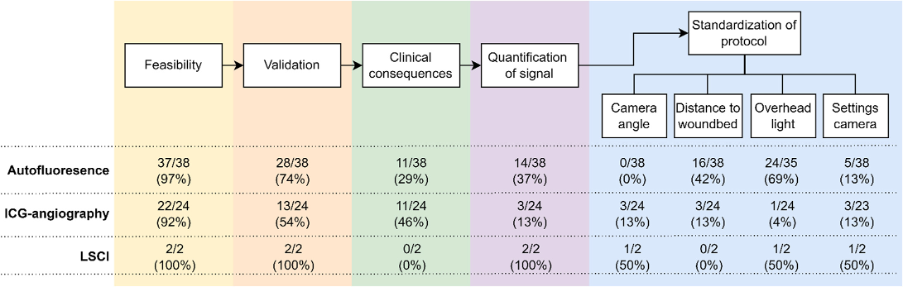
In another recent study of our group, we therefore started to design a standardization and quantification model for the fluorescence signal in ICG angiography, the WISQ model. The purpose of this is that the model can be used uniformly, independent of the user. In this study, 10 patients were included who underwent a total thyroidectomy, in which the parathyroid glands were imaged at the end of the procedure with ICG administration. In this model, attention was paid to the standardization of camera angle, distance to the wound bed, the camera settings and the fact that the overhead light was off. Subsequently, the ICG curves were analysed. In particular, the inflow and outflow slope of the best parathyroid gland per patient appeared to be predictive of postoperative hypoparathyroidism or no postoperative hypoparathyroidism. [5] (Figure 2)
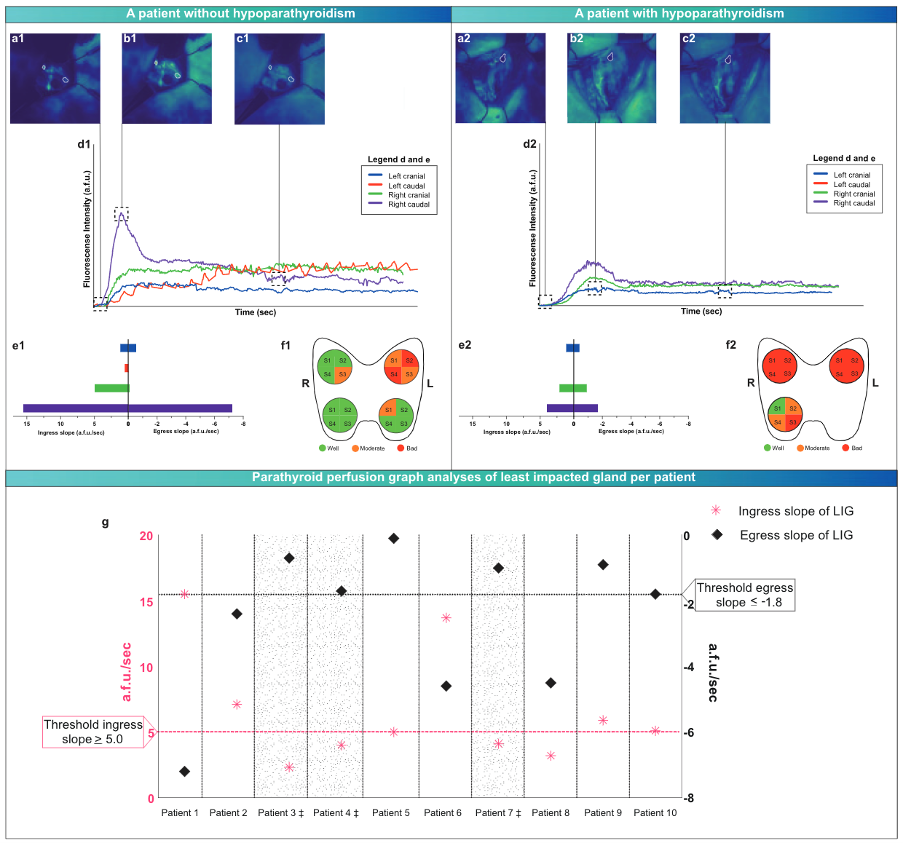
Study design
In this current multicenter phase 2 study, the aim is to validate the WISQ model in a larger group of patients (n=66). All patients undergoing total thyroidectomy can be included for this purpose, provided they have not previously had surgery in the neck region and do not have an iodine allergy. At the end of the procedure, images of the wound are taken with the fluorescence camera, with a standardized dose of ICG being administered. Furthermore, the camera is positioned and set up in the same way for each patient and all operating lights are turned off when the images are made. Postoperatively, the images are analyzed and perfusion curves are generated. From these perfusion curves, various parameters are determined, after which a correlation is made with the postoperative outcome.
This study is expected to be completed in the fall of 2022.
References
[1] Lonˇcar, I.; Noltes, M.E.; Dickhoff, C.; Engelsman, A.F.; Schepers, A.; Vriens, M.R.; Bouvy, N.D.; Kruijff, S.; van Ginhoven, T.M. Persistent Postthyroidectomy Hypoparathyroidism in the Netherlands. JAMA Otolaryngol.-Head Neck Surg. 2021, 147, 959–965.
[2] Edafe, O.; Antakia, R.; Laskar, N.; Uttley, L.; Balasubramanian, S.P. Systematic Review and Meta-Analysis of Predictors of Post-Thyroidectomy Hypocalcaemia. Br. J. Surg. 2014, 101, 307–320.
[3] Su, A.; Gong, Y.;Wu,W.; Gong, R.; Li, Z.; Zhu, J. Effect of Autotransplantation of a Parathyroid Gland on Hypoparathyroidism after Total Thyroidectomy. Endocr. Connect. 2018, 7, 286–294.
[4] Feitsma, E.A.; Schouw, H.M.; Noltes, M.E.; Heeman, W.; Kelder, W.; van Dam, G.M.; Kruijff, S. Heterogeneity in Utilization of Optical Imaging Guided Surgery for Identifying or Preserving the Parathyroid Glands—A Meta-Narrative Review. Life 2022, 12, 388.
[5] Noltes, M.E.; Metman, M.J.H.; Heeman, W.; Rotstein, L.; van Ginhoven, T.M.; Vriens, M.R.; Engelsman, A.F.; Boerma, C.E.; Brouwers, A.H.; van Dam, G.M.; et al. A Novel and Generic Workflow of Indocyanine Green Perfusion Assessment Integrating Standardization and Quantification Towards Clinical Implementation. Ann. Surg. 2021, 274, e659–e663.
